Light-Powered Reaction Creates Advanced Hybrid Nanomaterial
Exploring New Frontiers in Light-Driven Chemical Reactions
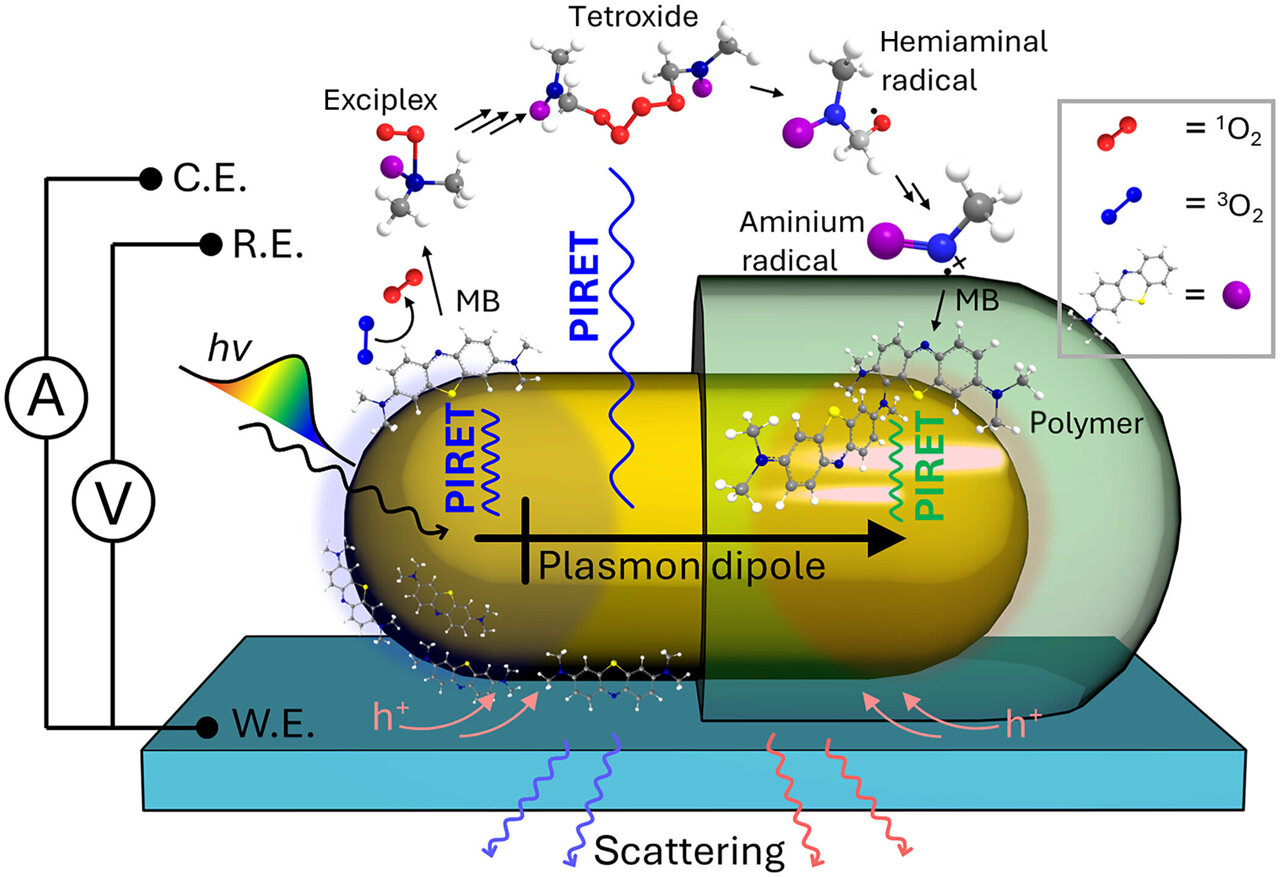
Scientists are increasingly looking for ways to harness light instead of heat to drive chemical reactions. This shift could lead to more efficient processes, reducing waste, energy use, and dependency on nonrenewable resources. One promising approach is plasmon-induced resonance energy transfer (PIRET), which involves transferring energy from a tiny metal particle to a semiconductor or molecule without direct contact.
At the University of Illinois Urbana-Champaign, a team of chemistry researchers has been investigating PIRET. Their work focuses on using light more efficiently, particularly through plasmonic metal nanoparticles, which are highly effective at absorbing and scattering light. This research is led by Christy Landes, an Illinois chemistry professor, who emphasizes the importance of optimizing light usage in chemical processes.
In a paper published in Science Advances, the team demonstrates how energy from a gold nanorod can be transferred to a synthetic blue dye, converting light into different forms until it becomes part of a polymer hybrid. This proof-of-concept study highlights PIRET's potential to revolutionize light-driven chemical reactions and create advanced hybrid nanomaterials in a way that differs from traditional charge-transfer catalysis.
"We can do a new type of polymerization chemistry that goes through a completely different intermediate route than the bulk polymerization reaction," Landes explains. "It doesn't happen the same way as a heat or pressure-initiated reaction, and that's a really special part of this work."
Stephan Link, another co-leader of the research team, adds that the method's potential for creating new reactions and materials is equally exciting. "We're learning the mechanisms behind it so that potentially we could design reactions that are otherwise not possible," he says.
Despite its promise, PIRET faces several challenges, including maximizing energy transfer efficiency and understanding its mechanism. The team addressed these issues by applying various methods to explore their specific PIRET-assisted polymerization reaction.
They used in-operando single-particle spectroelectrochemistry, a technique that allows real-time observation of energy transfer at the nanoscale. Their findings were supported by spectroscopic data, electrochemistry, and density functional theory calculations. Through this combination, they detailed a cascading energy transfer process from photon to plasmon to exciton, leading to a unique, light-initiated chemical transformation.
"We showed all pieces of that chain—our particles can absorb light very strongly; the energy can be transferred out very efficiently into molecules outside the nanoparticle; and we can do a new type of polymerization chemistry," Landes explains.
According to the study, the PIRET-assisted reaction demonstrated energetic spontaneity and worked at a lower energy level than traditional electropolymerization. They also reported a maximum PIRET efficiency of 40%, highlighting its potential in next-generation photocatalytic systems.
"I think that was an important demonstration that we've gone with a reaction that's known, but shown how this gives us a different, non-equilibrium route by taking out the energy from the particles through energy transfer to do the chemistry," Link says.
This research evolved from previous work by Hyuncheol Oh, a postdoctoral researcher in the Landes group. Initially studying energy transfer between a nanoparticle and methylene blue, he discovered he could initiate oligomerization, the first step toward a polymer. This finding marked the beginning of a new exploration into PIRET's capabilities.
"He really dug into the chemistry and the mechanism of this reaction, and I think a bunch of credit needs to go to him. He's been very passionate about this," Link notes.
Since then, Link has made a polymer, figured out the chemistry, and shown how light is the critical factor. He also identified singlet oxygen as a reactive intermediate that is very important.
The researchers believe this work deepens the understanding of PIRET mechanisms and opens new avenues for designing efficient, light-driven materials and reactions. Link sees great potential in this area.
"I think that could really open up the idea of using photons for catalysis and for different ways of doing chemistry. To me, that's something that could open up possibilities we're not even thinking about yet," he says.
Looking ahead, the team plans to explore other polymerization reactions using this approach. "This is a polymerization reaction that happens through a super complicated multi-step process, so this demonstrates that you can do really complicated chemistry using nanoparticles as light harvesters and for antennas to do the reactions. So definitely, we'd like to try some more complicated polymerization reactions," Landes says.
Other contributors to this work include Subhojyoti Chatterjee, Zhenyang Jia, Eric Gomez, Stephen A. Lee, Jiamu Lin, and Ojasvi Verma.
Post a Comment for "Light-Powered Reaction Creates Advanced Hybrid Nanomaterial"
Post a Comment